DESMODA helps bring precision, control,
and flexibility to CDI management1-8

DESMODA, the first and only FDA-approved oral solution of desmopressin for CDI, also known as AVP-D1,9
Limitations of Use: DESMODA is not indicated for the treatment of nephrogenic diabetes insipidus1
Antidiuretic replacement therapy with desmopressin, a synthetic analogue of AVP, is the standard of care for CDI10
The Society for Endocrinology recommends desmopressin as a first-line treatment for CDI.11
Desmopressin is distinct from endogenous AVP due to its:
- Long half-life (2+ hours, depending on formulation and individual)10,12
- Selectivity for the V2 receptor10
- Availability across multiple formulations10
Desmopressin is a synthetic analogue of AVP3
- The free amino group at position 1 is replaced by hydrogen
- The L-arginine at position 8 is replaced with D-arginine
- These chemical changes increase the diuretic effect and reduce the pressor effect of desmopressin relative to AVP
Clinical priorities of CDI management13
Control hallmark symptoms
The primary goal of treatment is to reduce polyuria and polydipsia
Prevent treatment-related hyponatremia
Therapy must balance water reabsorption and excretion to avoid excess water retention and subsequent hyponatremia
Address underlying causes when feasible
CDI may result from conditions such as head trauma, pituitary surgery, or tumors; treating the underlying cause may improve or restore AVP production
Use the lowest effective dose
Long-term management focuses on individualized titration to achieve symptom control while minimizing adverse effects

Precise desmopressin dosing is essential for effective CDI management14
- Insufficient desmopressin may result in hypernatremia due to excessive water clearance14
- Excessive desmopressin may result in hyponatremia due to excessive water retention14
Certain patient types are especially impacted by dosing variability
Infants and toddlers have wide variation in dose requirements and need frequent titration16
Patients with partial CDI and those with evolving disease may need to titrate doses as disease progresses17
Micro-variability in dosing can translate into macro-variability in clinical outcomes15
Current desmopressin formulations may have clinical limitations
Splitting tablets for patients who need low doses or complex titration may lead to dosing variability2,18
Many young children—and those with neuromotor difficulties—struggle with swallowing tablets8,19
Nasal sprays may deliver inconsistent doses if nasal congestion or inflammation is present3
Compounded formulations are not FDA-approved and are not clinically evaluated for safety, efficacy, or quality4
Injections may be associated with discomfort, needle fear, and injection site reactions5-7
CDI management involves serious monitoring burden and may lead to patient and caregiver uncertainty14
"Good enough" is no longer good enough
DESMODA, the first and only FDA-approved oral solution of desmopressin, helps enable precision, control, and flexibility across all ages when managing CDI1-8
- Formulation: Oral solution, 50 mcg/mL (0.05 mg/mL) concentration (1 mg=1000 mcg)
- Dosing precision: For noninjectable formulations, helps enable titrated and individualized dosing without tablet splitting or compounding. Provides tailored dosing using the minimal dose necessary (as low as 0.05 mg) to achieve the desired therapeutic effect
- Bioequivalence: Clinically demonstrated to be bioequivalent to desmopressin acetate tablets
- Ease of use: No refrigeration and no shaking required
- Administration benefits: Eliminates the need for subcutaneous injections or intranasal administration
- Access model: Exclusively available through a specialty pharmacy with a comprehensive patient support program, including $0 copay for eligible patients

DESMODA helps empower you to treat CDI with control
Deliver trusted CDI treatment with flexibility and control1
Effective CDI management depends on individualized, carefully tailored dosing10,16,21
For all patients, DESMODA dosing must be personalized and adjusted according to the diurnal pattern of response.1
In pediatric patients & adults1,22
Recommended starting dose is 0.05 mg (1 mL of solution) 2 times a day. Adjust to the patient’s optimum therapeutic dose.
- Each dose should be separately adjusted for an adequate diurnal rhythm of water turn over
- Titrate the daily dosage as needed to obtain an adequate antidiuretic response
- Total daily dosage should be increased or decreased in the range of 0.1 mg to 1.2 mg divided into 2 or 3 daily doses as needed to obtain adequate antidiuresis
- Any unused DESMODA should be discarded after 120 days from first opening the bottle
In adult dose–response studies15,22
Oral desmopressin doses from 0.025 mg to 0.4 mg provided significant antidiuretic effects in patients with CDI.
- For most patients, doses of 0.1–0.2 mg produce optimal antidiuretic effects lasting up to 8 hours
Pediatric dosing may be less than 0.1–0.2 mg
- A 0.4 mg dose can produce antidiuretic effects for up to 12 hours (measurements beyond 12 hours were not recorded)
In younger pediatric patients1,23
Dosing should start at 0.05 mg. Individually adjust dose to prevent excessive decrease in plasma osmolality, which can lead to hyponatremia and convulsions.
- Use careful fluid intake restrictions to prevent hyponatremia and water intoxication
- Fluid restriction should be discussed with the patient and/or guardian
- DESMODA is taste-neutral with no added flavors, which may have a positive impact on pediatric adherence
Patients previously on intranasal desmopressin acetate therapy should begin solution therapy 12 hours after the last intranasal dose.1
Monitoring and observing patients receiving DESMODA1
Patients should be closely monitored during initial dose titration and at regular intervals during therapy to ensure an adequate antidiuretic response.
- Modifications in the dosing regimen should be implemented as necessary to ensure adequate water turnover
- Fluid restriction should be observed during therapy

Intake guidelines1
DESMODA should be taken only on an empty stomach
(1 hour before or 2 hours after a meal)
- In a food-effect study, DESMODA showed decreased absorption after a high-fat meal
- In a different study, desmopressin acetate tablets also demonstrated a significant food effect20

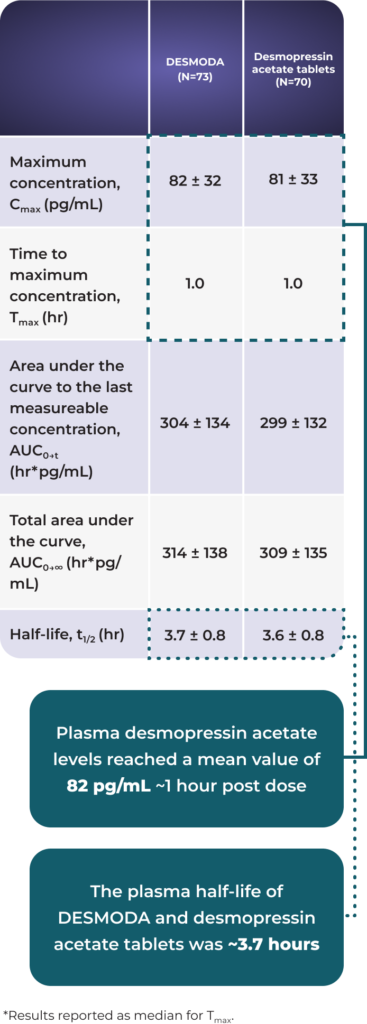
Demonstrated bioequivalence to desmopressin acetate tablets20
In an open-label bioequivalence study, DESMODA showed similar pharmacokinetic results as desmopressin acetate tablets20
Pharmacokinetic comparison: DESMODA vs tablets (mean ± SD)20*

Safety data
DESMODA offers comparable pharmacokinetics to tablets paired with the precision of an oral solution1,20

Desmopressin acetate has a demonstrated safety and tolerability profile among pediatric and adult patients1,20*
All patients should report symptoms of hyponatremia. See “Hyponatremia risk” section below.1
In patients younger than 4 years of age, the dose must be individually adjusted to prevent an excessive decrease in plasma osmolality leading to hyponatremia and possible convulsions; dosing should start at 0.05 mg.1
DESMODA is not indicated for the treatment of nephrogenic diabetes insipidus.1

Safety data1
Most common adverse reactions
- Headache, dizziness, nausea, abdominal pain, hypertension, and diarrhea
Clinical trials experience
In long-term clinical studies in which patients with CDI were followed for periods up to 44 months of desmopressin acetate oral therapy, transient increases in AST (SGOT) ≤1.5x the upper limit of normal were occasionally observed- Elevated AST (SGOT) returned to the normal range despite the continued use of desmopressin acetate oral therapy
Postmarketing experience
There have been rare reports of hyponatremic convulsions associated with concomitant use of oxybutynin and imipramine.Drug interactions
- Although the pressor activity of desmopressin acetate is very low compared to its antidiuretic activity, large doses of DESMODA should be used with other pressor agents only with careful patient monitoring
- Concomitant administration of drugs that may increase the risk of water intoxication with hyponatremia (eg, tricyclic antidepressants, selective serotonin re-uptake inhibitors, chlorpromazine, opiate analgesics, NSAIDs, lamotrigine, and carbamazepine) should be performed with caution
Additional safety considerations1
DESMODA should not be used in patients who have:
- Hyponatremia or a history of hyponatremia
- Hypersensitivity to desmopressin acetate or to any of the inactive ingredients of DESMODA. Reactions have included anaphylaxis
- Moderate to severe renal impairment (defined as a creatinine clearance below 50 mL/min)
Situations requiring close clinical oversight:
- Careful monitoring should be observed when large doses of DESMODA are used with other pressor agents or when co-administering DESMODA with drugs that increase the risk of water intoxication and hyponatremia
- Closely monitor patients with heart failure or uncontrolled hypertension, as desmopressin acetate can cause fluid retention, which can worsen underlying conditions that are susceptible to volume status
– DESMODA is not recommended in patients at risk for increased intracranial pressure or those with a history of urinary retention
Hyponatremia risk1
Postmarketing experience
Very rare cases of hyponatremia have been reported from worldwide postmarketing experience in patients treated with DESMODA.
- DESMODA is a potent antidiuretic that, when administered, may lead to water intoxication and/or hyponatremia
- Unless properly diagnosed and treated, hyponatremia can be fatal
- Therefore, fluid restriction is recommended and should be discussed with the patient and/or guardian
Fluid intake
- When DESMODA is administered, particularly in pediatric and geriatric patients, fluid intake should be adjusted downward to decrease the potential occurrence of water intoxication and hyponatremia
- All patients receiving DESMODA should be monitored for the following signs of symptoms associated with hyponatremia:
– Headache
– Nausea/vomiting
– Decreased serum sodium
osmolality
– Weight gain
– Restlessness
– Fatigue
– Lethargy
– Disorientation
– Depressed reflexes
– Loss of appetite
– Irritability
– Muscle weakness
– Muscle spasms or cramps
– Abnormal mental status such
as hallucinations, decreased
consciousness, and confusion
- Particular attention should be paid to the possibility of the rare occurrence of an extreme decrease in plasma osmolality that may result in seizures, which could lead to coma
Polydipsia
- DESMODA should be used with caution in patients with habitual or psychogenic polydipsia who may be more likely to drink excessive amounts of water, putting them at greater risk of hyponatremia

DESMODA is approved for patients of all ages1

Patient profiles
Potential patient scenarios where DESMODA may be considered
Patient profiles are fictional and are intended for illustrative purposes only.
Olivia, age 1
A case where tablet dosing was not a viable option and treatment with nasal sprays was unsuccessful
Patient history
- Olivia suffered from polyuria/polydipsia as an infant
- Her required dose was too small for whole tablets, and treatment with desmopressin nasal spray proved unreliable
– Caregivers were concerned that dosing inconsistency resulting from splitting tablets could lead to underdosing or overdosing
– Frequent colds and nasal infections interfered with absorption
Transition to DESMODA
- Olivia now receives DESMODA, allowing for precise, individualized dose titration in an oral solution, designed to help reduce dosing complexity and support more predictable dosing over time1-3
Liam, age 8
A case where tablet dosing was not a viable option
Patient history
- Liam developed CDI after a traumatic brain injury
- Motor control challenges resulting from the injury contributed to dysphagia, making oral tablet administration difficult
- His caregivers crushed desmopressin tablets to facilitate administration, but were concerned about inconsistent doses
- Parenteral administration proved unsustainable due to the burden of injection site reactions and needle fear
Transition to DESMODA
- Liam is now prescribed DESMODA, providing a precise oral solution dosing option that is designed to support greater control and clarity in ongoing management1,6,7
Noah, age 12
A case where partial CDI required steadily increasing doses over time
Patient history
- Noah was diagnosed with partial CDI at age 11
- Displayed mild polydipsia/polyuria symptoms from age 6, but was misdiagnosed for years
- Began treatment with desmopressin tablets; however, his increasing symptom severity necessitated constant titration
- Caregivers were concerned that tablets would not offer precise dosing increments
Transition to DESMODA
- Noah transitioned to DESMODA, offering a precise oral solution dosing approach that helps caregivers manage changing treatment titration needs1,2
DESMODA is appropriate for patients of all ages, including1:
- Pediatric patients
- Patients with administration challenges from tablet or nasal dosage forms
- Patients with partial CDI
Which patients in your practice would benefit from DESMODA?

Access DESMODA patient support and resources for you and your practice
References: 1. DESMODA. Package Insert. Eton Pharmaceuticals; 2026. 2. Verrue C, Mehuys E, Boussery K, Remon JP, Petrovic M. Tablet-splitting: a common yet not so innocent practice. J Adv Nurs. 2011;67(1):26-32. 3. Chin X, Teo SW, Lim ST, Ng YH, Han HC, Yap F. Desmopressin therapy in children and adults: pharmacological considerations and clinical implications. Eur J Clin Pharmacol. 2022;78(6):907-917. 4. Gudeman J, Jozwiakowski M, Chollet J, Randell M. Potential risks of pharmacy compounding. Drugs R D. 2013;13(1):1-8. 5. Zhi L, Liu D, Shameem M. Injection site reactions of biologics and mitigation strategies. AAPS Open. 2025;11:5. 6. McLenon J, Rogers MAM. The fear of needles: A systematic review and meta-analysis. J Adv Nurs. 2019;75(1):30-42. 7. Centers for Disease Control and Prevention. Before, during, and after shots. CDC. August 9, 2024. Accessed February 2, 2026. https://www.cdc.gov/vaccines-children/before-during-after-shots/index.html 8. VandenBerg CJ, Adams A, Bockrath R, et al. Hard to swallow: a review of interventions to improve swallowing solid medication. Hosp Pediatr. 2023;13(5):e123-e132. 9. Arima H, Cheetham T, Christ-Crain M, et al. Changing the name of diabetes insipidus: a position statement of the Working Group for Renaming Diabetes Insipidus. J Clin Endocrinol Metab. 2022;108(1):1-3. 10. Atila C, Refardt J, Christ-Crain M. Arginine vasopressin deficiency: diagnosis, management and the relevance of oxytocin deficiency. Nat Rev Endocrinol. 2024;20(8):487-500. 11. Baldeweg SE, Ball S, Brooke A, et al. Society for Endocrinology Clinical Guidance: inpatient management of cranial diabetes insipidus. Endocr Connect. 2018;7(7):G8-G11. 12. Kalra S, Zargar AH, Jain SM, et al. Diabetes insipidus: The other diabetes. Indian J Endocrinol Metab. 2016;20(1):9-21. 13. Tomkins M, Lawless S, Martin-Grace J, Sherlock M, Thompson CJ. Diagnosis and management of central diabetes insipidus in adults. J Clin Endocrinol Metab. 2022;107(10):2701-2715. 14. Teare H, Argente J, Dattani M, et al. Challenges and improvement needs in the care of patients with central diabetes insipidus. Orphanet J Rare Dis. 2022;17(1):58. 15. Yen K, Hughes E, Savic R, Srinivasan S. Desmopressin dosing in children using real-world data and pharmacokinetic/pharmacodynamic model simulations. Pediatr Res. 2025;97(7):2449-2453. 16. Ooi HL, Maguire AM, Ambler GR. Desmopressin administration in children with central diabetes insipidus: a retrospective review. J Pediatr Endocrinol Metab. 2013;26(11-12):1047-1052. 17. Pedersen AN, Andreassen M, Rasmussen AK, Krogh J. Desmopressin dose requirements in adults with congenital and acquired central diabetes insipidus. Horm Metab Res. 2024;56(3):206-213. 18. Saran AK, Holden NA, Garrison SR. Concerns regarding tablet splitting: a systematic review. BJGP Open. 2022;6(3):BJGPO.2022.0001. 19. van den Engel-Hoek L, de Groot IJ, de Swart BJ, Erasmus CE. Feeding and swallowing disorders in pediatric neuromuscular diseases: an overview. J Neuromuscul Dis. 2015;2(4):357-369. 20. Data on file. Eton Pharmaceuticals; March 2025. 21. Christ-Crain M, Bichet DG, Fenske WK, et al. Diabetes insipidus. Nat Rev Dis Primers. 2019;5(1):54. 22. Desmopressin acetate tablets. Package Insert. Ferring Pharmaceuticals Inc; 2019. 23. Ranmal SR, Walsh J, Tuleu C. Poor-tasting pediatric medicines: Part 1. A scoping review of their impact on patient acceptability, medication adherence, and treatment outcomes. Front Drug Deliv. 2025;5:1553286.
USE AND IMPORTANT SAFETY INFORMATION
INDICATION
DESMODA (desmopressin acetate) is a vasopressin analog indicated for the management of central diabetes insipidus as antidiuretic replacement therapy for adults and pediatric patients.
Limitations of Use
Do not use DESMODA for the treatment of nephrogenic diabetes insipidus.
IMPORTANT SAFETY INFORMATION
Contraindications
DESMODA is contraindicated in patients with hypersensitivity to desmopressin acetate or to any of the components of DESMODA, patients with moderate to severe renal impairment (adults with creatinine clearance (CLcr) less than 50 mL/min), or patients with hyponatremia or a history of hyponatremia.
Warnings and Precautions
Hyponatremia: Excessive fluid intake when urine output is limited by the antidiuretic effect of desmopressin may lead to water intoxication with hyponatremia. Cases of hyponatremia have been reported from postmarketing experience with desmopressin acetate. Monitor patients for signs or symptoms associated with hyponatremia, including headache, nausea/vomiting, weight gain, restlessness, fatigue, lethargy, confusion, depressed reflexes, muscle cramps or spasms, and abnormal mental status. Severe hyponatremia may result in seizures, coma, respiratory arrest, or death.
Fluid restriction is recommended during treatment and is particularly important in pediatric and geriatric patients, who are at increased risk. More frequent monitoring of serum sodium is recommended in patients with conditions associated with fluid and electrolyte imbalance or those receiving concomitant medications that may cause hyponatremia. Temporarily stop treatment with DESMODA during acute intercurrent illness characterized by fluid and/or electrolyte imbalance or under conditions associated with increased water intake.
Fluid Retention: Desmopressin acetate may cause fluid retention and should be used with caution in patients with heart failure or uncontrolled hypertension. DESMODA is not recommended in patients at risk for increased intracranial pressure or those with a history of urinary retention.
Hypersensitivity Reactions: Hypersensitivity reactions including anaphylaxis have been reported rarely with intravenous and nasal administration of desmopressin acetate. DESMODA is contraindicated in patients with known hypersensitivity to desmopressin acetate or any of the components of DESMODA.
Risk of Benzyl Alcohol Toxicity in Neonates: Serious adverse reactions, including fatal reactions, have been reported in low-birth-weight neonates and preterm neonates who received benzyl alcohol containing drugs intravenously. DESMODA contains benzoic acid, a metabolite of benzyl alcohol; the relationship between systemic benzoic acid exposure and toxicity is not well characterized. Use DESMODA with caution in low-birth-weight neonates or preterm neonates and monitor for signs and symptoms of metabolic acidosis.
Adverse Reactions
The serious adverse reactions associated with DESMODA are hyponatremia, fluid retention, hypersensitivity, and the risk of benzyl alcohol toxicity in neonates. Other common adverse reactions reported with desmopressin acetate include abnormal thinking, diarrhea, and edema/weight gain. Additional adverse reactions reported in clinical studies or postmarketing experience include nausea, vomiting, headache, fatigue, dizziness, water intoxication, seizures, confusion, hallucinations, urinary retention, and rash.
To report a suspected adverse event related to DESMODA, contact Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at https://www.fda.gov/safety/medwatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.
USE AND IMPORTANT SAFETY INFORMATION
INDICATION
DESMODA (desmopressin acetate) is a vasopressin analog indicated for the management of central diabetes insipidus as antidiuretic replacement therapy for adults and pediatric patients.
Limitations of Use
Do not use DESMODA for the treatment of nephrogenic diabetes insipidus.
IMPORTANT SAFETY INFORMATION
Contraindications
DESMODA is contraindicated in patients with hypersensitivity to desmopressin acetate or to any of the components of DESMODA, patients with moderate to severe renal impairment (adults with creatinine clearance (CLcr) less than 50 mL/min), or patients with hyponatremia or a history of hyponatremia.
Warnings and Precautions
Hyponatremia: Excessive fluid intake when urine output is limited by the antidiuretic effect of desmopressin may lead to water intoxication with hyponatremia. Cases of hyponatremia have been reported from postmarketing experience with desmopressin acetate. Monitor patients for signs or symptoms associated with hyponatremia, including headache, nausea/vomiting, weight gain, restlessness, fatigue, lethargy, confusion, depressed reflexes, muscle cramps or spasms, and abnormal mental status. Severe hyponatremia may result in seizures, coma, respiratory arrest, or death.
Fluid restriction is recommended during treatment and is particularly important in pediatric and geriatric patients, who are at increased risk. More frequent monitoring of serum sodium is recommended in patients with conditions associated with fluid and electrolyte imbalance or those receiving concomitant medications that may cause hyponatremia. Temporarily stop treatment with DESMODA during acute intercurrent illness characterized by fluid and/or electrolyte imbalance or under conditions associated with increased water intake.
Fluid Retention: Desmopressin acetate may cause fluid retention and should be used with caution in patients with heart failure or uncontrolled hypertension. DESMODA is not recommended in patients at risk for increased intracranial pressure or those with a history of urinary retention.
Hypersensitivity Reactions: Hypersensitivity reactions including anaphylaxis have been reported rarely with intravenous and nasal administration of desmopressin acetate. DESMODA is contraindicated in patients with known hypersensitivity to desmopressin acetate or any of the components of DESMODA.
Risk of Benzyl Alcohol Toxicity in Neonates: Serious adverse reactions, including fatal reactions, have been reported in low-birth-weight neonates and preterm neonates who received benzyl alcohol containing drugs intravenously. DESMODA contains benzoic acid, a metabolite of benzyl alcohol; the relationship between systemic benzoic acid exposure and toxicity is not well characterized. Use DESMODA with caution in low-birth-weight neonates or preterm neonates and monitor for signs and symptoms of metabolic acidosis.
Adverse Reactions
The serious adverse reactions associated with DESMODA are hyponatremia, fluid retention, hypersensitivity, and the risk of benzyl alcohol toxicity in neonates. Other common adverse reactions reported with desmopressin acetate include abnormal thinking, diarrhea, and edema/weight gain. Additional adverse reactions reported in clinical studies or postmarketing experience include nausea, vomiting, headache, fatigue, dizziness, water intoxication, seizures, confusion, hallucinations, urinary retention, and rash.
To report a suspected adverse event related to DESMODA, contact Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at https://www.fda.gov/safety/medwatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.